Complete the following reactions:
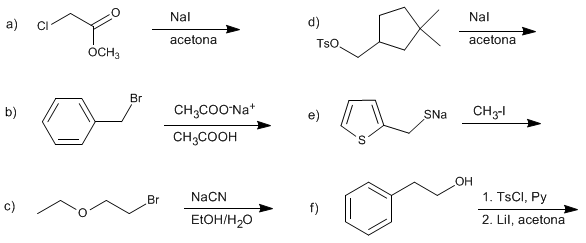
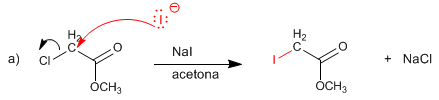
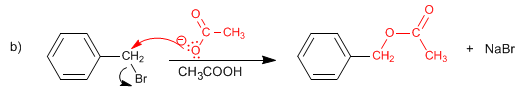
SOLUTION:
The iodide ion acts as a nucleophile, replacing the chloride.
The acetate ion replaces bromine by the SN2 mechanism.
The cyanide ion acts as a nucleophile, replacing the bromine.
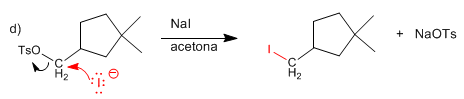
Tosyl is replaced by the SN2 mechanism by iodide.
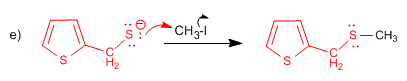
The sulfur compound attacks the carbon of the methyl iodide, replacing the iodine.
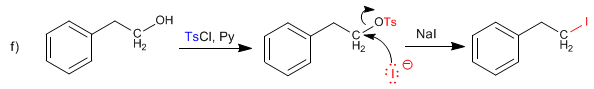
This last reaction occurs in two stages. In the first, a good leaving group is obtained by using tosyl chloride. In the second stage, the replacement of the tosylate by iodide occurs.