Rule 1. The longest chain that contains the -OH group is chosen as the main chain.

Rule 2. The main chain is numbered so that the -OH group takes the lowest locant. The hydroxyl group has preference over carbon chains, halogens, double and triple bonds.
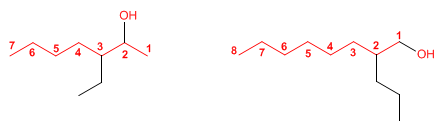
Rule 3. The name of the alcohol is constructed by changing the ending -o of the alkane with the same number of carbons to -ol
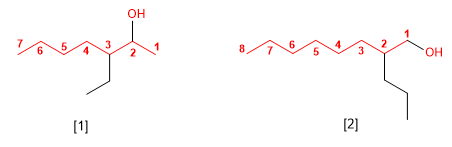
[1] 3-Ethylheptan-2-ol
[2] 2-Propyloctan-1-ol
Rule 4. When there are functional groups of higher priority in the molecule, the alcohol becomes a mere substituent and is called hydroxy- . They have priority over alcohols: carboxylic acids, anhydrides, esters, alkanoyl halides, amides, nitriles, aldehydes and ketones.
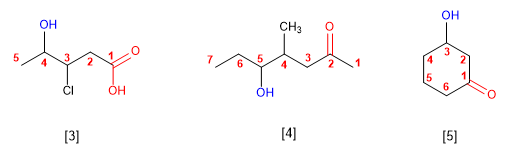
[3] 3-Chloro-4-hydroxypentanoic acid
[4] 5-Hydroxy-4-methylheptan-2-one
[5] 3-Hydroxycyclohexanone
Rule 5 . The -OH group has priority over alkenes and alkynes. The numbering gives the lowest locant to -OH and the name of the molecule ends in -ol.
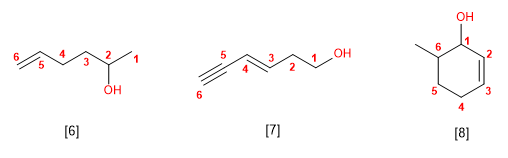
[6] Hex-5-en-2-ol
[7] Hex-3-in-5-in-1-ol
[8] 6-Methylcyclohex-2-en-1-ol