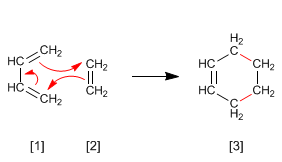
1,3-Butadiene (diene) adds to ethene (dienophile) to form cyclohexene. Diels-Alder forms six-membered cycles, which is why it is known as a cycloaddition reaction. The transition state of the reaction is cyclic, belonging to the family of pericyclic reactions.
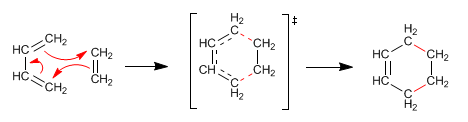
The Diels-Alder reaction is favored when one of the components has groups that transfer charge and the other groups that steal it.
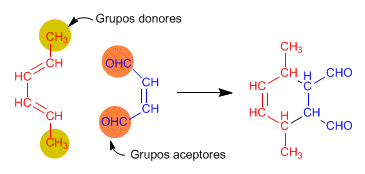
The Diels-Alder product always has one more cycle than the starting reactants