Cyanohydrins are formed by reaction of aldehydes or ketones with hydrocyanic acid and are compounds that contain a cynane and a hydroxy group on the same carbon. 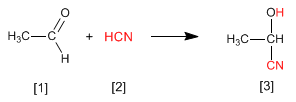
The reaction mechanism occurs in two stages:
Stage 1. Cyanide ions act as nucleophiles attacking the carbonyl carbon. Hydrocyanic acid is too weak to generate significant amounts of cyanide, therefore sodium or potassium cyanide is added to the medium, guaranteeing a sufficient amount of cyanide for the reaction to proceed in good yield. 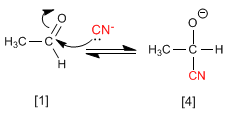
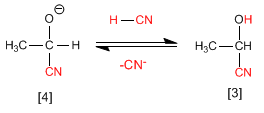
Stage 2. In this step the alkoxide ion it is protonated by removing hydrogens from hydrocyanic acid. In this stage, cyanide ions are regenerated.