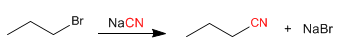
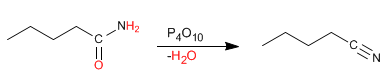a) From haloalkanes: Nitriles can be prepared from haloalkanes, by SN2 processes. The reaction gives good performance with primary and secondary substrates, the tertiary ones are preferentially eliminated, forming alkenes.
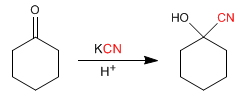
b) From carbonyls: Aldehydes and ketones react with hydrocyanic acid, forming cyanohydrins.
c) From amides: amides dehydrate to give nitriles. Powerful dehydrants are used, such as P 2 O 5 , which remove a water molecule from the amide, converting it to nitrile.