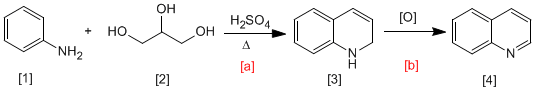
In the Skraup synthesis, aniline reacts with a,b-unsaturated carbonyls in an acid medium to form 1,2-dihydroquinolines, which are transformed into quinolines by oxidation. The a,b-unsaturated carbonyl can be obtained by dehydration of 1,2,3-propanetrium.
Mechanism:
In this synthesis we start with 1,2,3-propanetriol that is transformed into propenal by treatment with sulfuric acid. 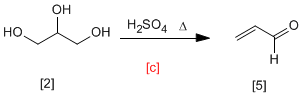
In the first stage of the mechanism, the Michael addition between the amine and the a,b -unsaturated occurs to form an enol that generates a b-aminocarbonyl after losing a proton and a ketoenol tautomerism. 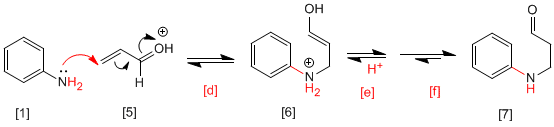
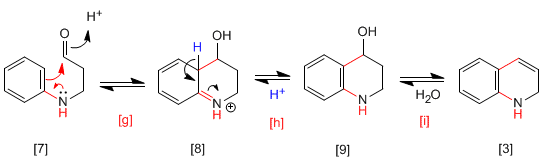
The compound cyclizes by attack of benzene, after protonation of the carbonyl, aided by the transfer of the non-bonding pair of nitrogen, forming the intermediate, which rearomatizes by loss of proton, generating the compound. Dehydration of alcohol produces 1,2-dihydroquinoline.