The anhydrides are obtained by condensation of carboxylic acids with loss of water. The reaction requires strong heating and a long reaction time.
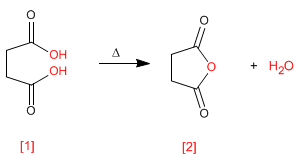
heating butanedioic acid [1] produces butanedioic anhydride (succinic anhydride) [2] . This type of cyclization requires 5- or 6-membered rings.
Obtaining anhydrides can be carried out by reaction of alkanoyl halides and carboxylic acids. In this case, milder conditions are required due to the significant reactivity of the alkanoyl halide.
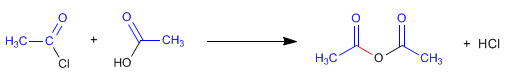
The mechanism of this reaction begins with the attack of the oxygen of the carboxylic group on the carbon of the alkanoyl halide, producing the elimination of chlorine in a second stage. The slow step of the mechanism is nucleophilic attack and can be favored by previously deprotonating the carboxylic acid.
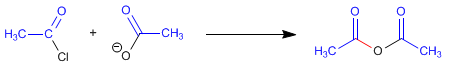
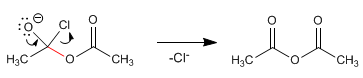
The carboxylate and alkanoyl halide react at room temperature to form the anhydride. The mechanism of the reaction occurs with the following stages:
Stage 1 . Addition of the carboxylate to the halide
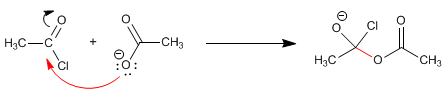
Stage 2. Elimination of chloride.