Acid derivatives, alkanoyl halides, anhydrides, esters, nitriles, and amides, show important differences in reactivity toward nucleophiles.
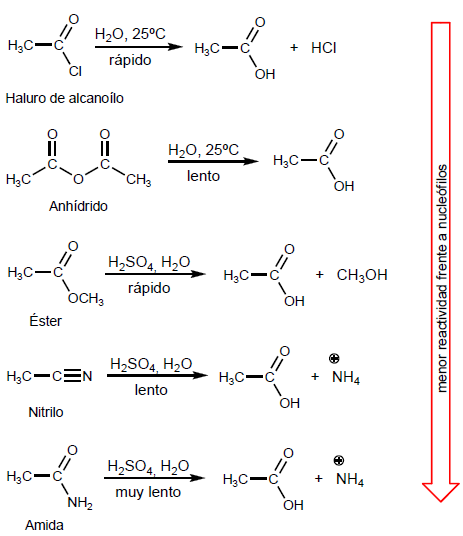
The order of reactivity of carboxylic acids is related to the ability of the L group to transfer charge.
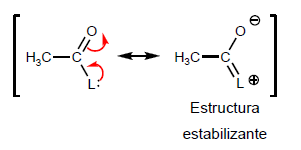
The greater the capacity of the L group to yield lone pairs, a lower reactivity is observed, due to the formation of a stabilizing limit structure.
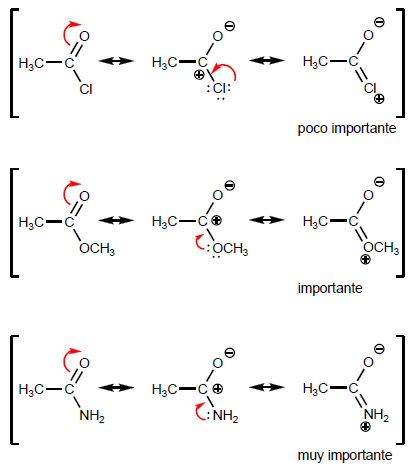
The greater the weight of the latter structure, the less reactivity the corresponding acid derivative possesses.