The Hofmann elimination allows the conversion of amines to alkenes. It is a regioselective reaction that follows Hofmann's rule, forming the least substituted alkene.
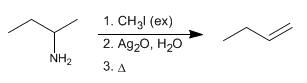
Stage 1. Exhaustive methylation of the amine. In this step the amine is reacted with excess methyl iodide, to form an ammonium salt (good leaving group).
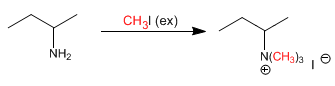
Stage 2. Treatment with aqueous silver oxide. Basic salt that forms an ammonium hydroxide, precipitating the iodide in the form of silver iodide.
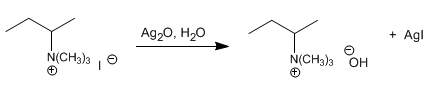
Step 3. Bimolecular elimination. Ammonium hydroxide undergoes an E2 on heating, which gives rise to the alkene.
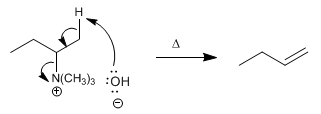
Being an E2, the Hofmann deletion has ANTI stereochemistry.