Rank the following alkenes in order of decreasing stability: a) 1-pentene; b) (E)-2-pentene; c) (Z)-2-pentene; d) 2-methyl-2-butene.
SOLUTION:
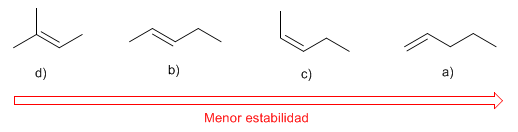
The stability of alkenes increases with the number of chains that start from the carbons of the double bond (the more stable the more substituted).
The alkene d) is the most stable because it has three substituents. b) is more stable than c) because it is a trans alkene. a) is the alkene with the least stability (it has only one substituent)