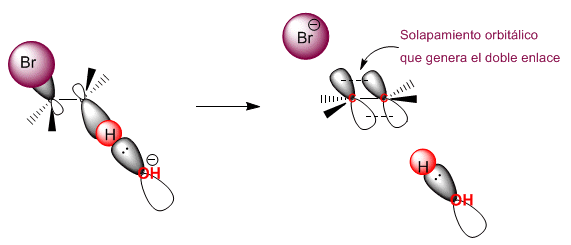
Bimolecular (E2) eliminations take place at a faster rate if the leaving group is on the opposite side of the removed hydrogen. This spatial arrangement is known as "ANTI"
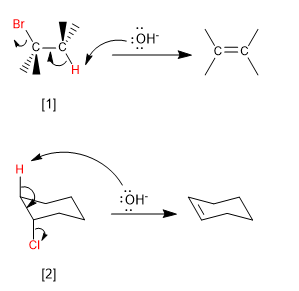
[1] H and Br in "ANTI"
[2] H and Cl in "ANTI"
The "ANTI" deletion is favored because it maximizes the overlap between the two nascent p orbitals giving rise to pi bond formation.
In the following example, the two b carbons have hydrogens, however, the base mostly removes the hydrogen that is ANTI with respect to chlorine (red hydrogen)
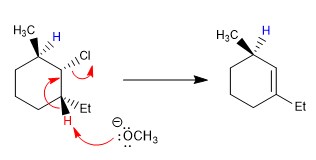
H hydrogen ANTI with respect to chlorine
H hydrogen SYN with respect to chlorine.