Name the following branched alkanes: 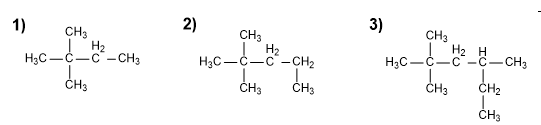
SOLUTION:
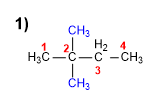
1. Main chain: the longest chain is chosen as the main chain. It will name the alkane (butane).
2. Numbering: it is numbered so that the substituents (methyls) take the minor locants.
3. Substituents: two methyls in position 2 . When there are two identical substituents, the prefix di- is used, preceded by the localizers, separated by commas.
4. Name: 2,2-Dimethylbutane
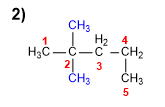
1. Main chain: The longest chain is five carbons (pentane).
2. Numbering: The numbering starts from the left to assign locant 2 to the methyls.
3. Substituents: two methyls in position 2.
4. Name: substituents preceded by their locants, ending with the name of the main chain. 2,2-Dimethylpentane.
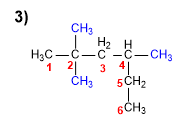
1. Main chain: in this case the main chain is not the horizontal one, which only has five carbons.
2. Numbering: start with the end that is closest to a substitute (left).
3. Substituents: The main chain contains three identical substituents (methyls), which are named using the prefix tri-, preceded by the locants indicating the position on the main chain ( 2,2,4 ).
4. Name: 2,2,4-Trimethylhexane