Name the following ethers:
SOLUTION:
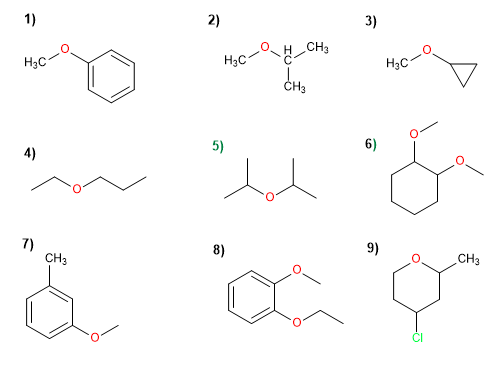
Molecule 1.
1. Substituents: phenyl and methyl
2. Name: Phenyl methyl ether
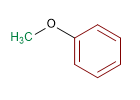
Molecule 2.
1. Substituents: isopropyl and methyl
2. Name: Isopropyl methyl ether
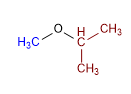
Molecule 3.
1. Substituents: cyclopropyl and methyl
2. Name: Cyclopropyl methyl ether
![]() The name of the ethers is built ending in the word ether the name of the chains that start from the oxygen. These chains are named as substituents and are arranged alphabetically. Note the space between the words.
The name of the ethers is built ending in the word ether the name of the chains that start from the oxygen. These chains are named as substituents and are arranged alphabetically. Note the space between the words.
Molecule 4.
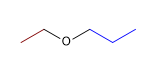
1. Substituents: ethyl and propyl
2. Name: Ethyl Propyl Ether
Molecule 5.
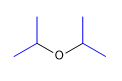
1. Substituents: isopropyls
2. Name: Diisopropyl ether
Molecule 6.
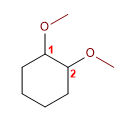
1. Main chain: six-membered cycle (cyclohexane)
2. Numbering: Give lower locants to substituents
3. Substituents: 1,2-methoxyde
4. Name: 1,2-Dimethoxycyclohexane
Molecule 7.
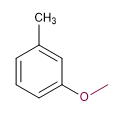
1. Main chain: Toluene
2. Numbering: methyl and meta methoxide.
3. Substituents: methoxide
4. Name: m-Methoxytoluene
Molecule 8.
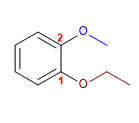
1. Main chain: Benzene
2. Numbering: Starts at the ethoxy (previously alphabetically)
3. Substituents: 1-ethoxyde and 2-methoxyde. (goal position)
4. Name: m-Ethoxymethoxybenzene
Molecule 9.
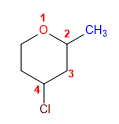
1. Main chain: 6-membered cycle (oxacyclohexane)
2. Numbering: Start at oxygen, proceed to the right to give substituents the minor locants.
3. Substituents: chlorine and methyl
4. Name: 4-Chloro-2-methyloxacyclohexane