Write the structure of the three alkenes that are obtained in the acid-catalyzed dehydration of 2-pentanol.
SOLUTION:
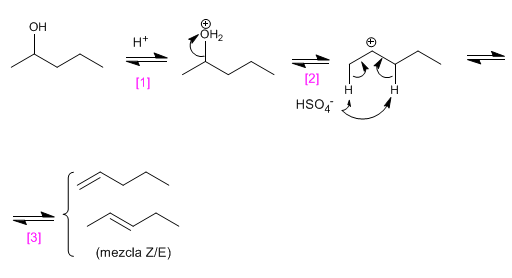
[1] Alcohol protonation.
[2] Loss of water, with formation of secondary carbocation.
[3] Elimination promoted by the bases of the environment.