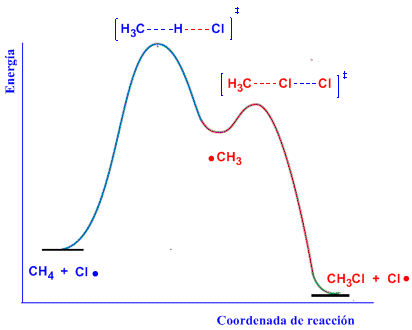
We are going to represent in an energy diagram the two stages of propagation of the halogenation of methane.
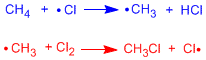
The first stage of propagation is the one that limits the speed of the process, it has the highest activation energy. The diagram represents reactants, products, intermediates, and transition states for the radical halogenation of methane.
In the first transition state the H-Cl bond is formed and the CH is broken. In the second transition state the C-Cl bond is formed and the Cl-Cl breaks.