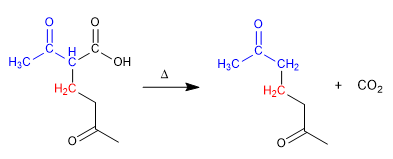
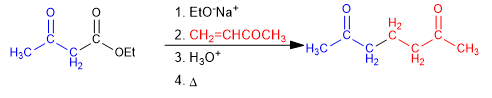
When the electrophile used in the acetylacetic or malonic synthesis is a,b -unsaturated, a 1,5-dicarbonyl will be formed, this type of reaction is called Michael addition.
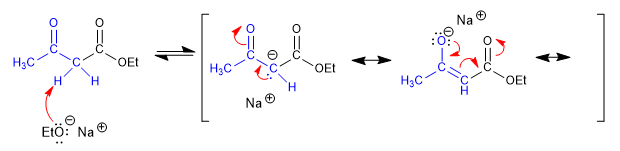
Stage 1. Formation of the ketoester enolate
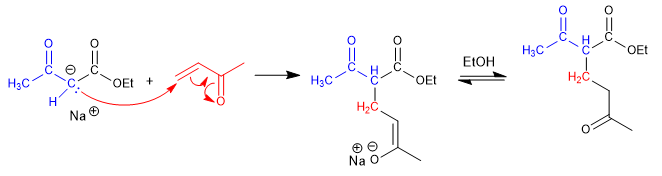
Step 2. Nucleophilic attack of the a,b -unsaturated ketoester enolate
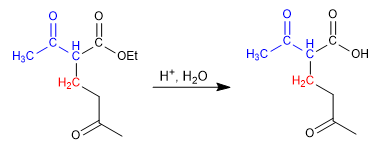
Stage 3. Hydrolysis of the ester
Stage 4. Decarboxylation