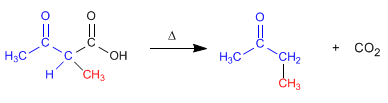Ethyl 3-oxobutanoate (ethyl acetylacetate), can be obtained by Claisen from ethyl acetate, and is a very efficient reagent for the preparation of ketones.
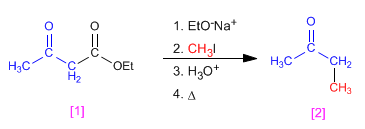
Butanone [2] can be obtained from ethyl acetylacetate [1] through the indicated stages.
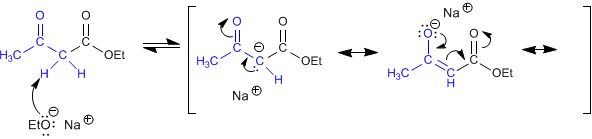
Stage 1. Formation of the ketoester enolate
Stage 2 . Nucleophilic attack of the ketoester enolate on the haloalkane.
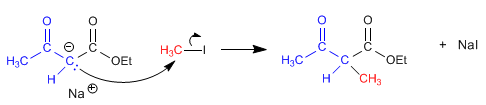
Stage 3. Hydrolysis of the ester
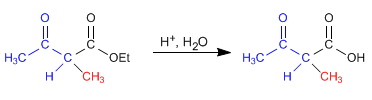
Stage 4. Decarboxylation