Amino acids can be obtained by halogenation of carboxylic acids, followed by nucleophilic substitution with ammonia. The halogenation of the a position of the carboxylic acid is carried out with the Hell-Volhard-Zelinsky reaction.
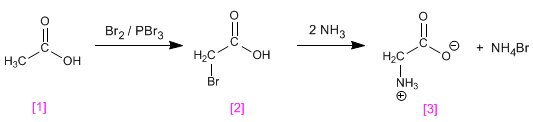
wisteria can be prepared from ethanoic acid . In the first stage, ethanoic acid reacts with phosphorus-catalyzed bromine to form the halogenated acid at its a position . The reaction of with two equivalents of ammonia produces Glycine through SN 2 plus ammonium bromide.