The different spatial arrangements that a molecule can adopt and that are interconverted at room temperature by rotation are called conformations.
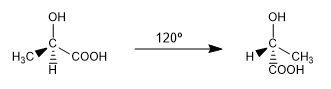
They are two of the infinite conformations that can be drawn from the ac. 2-hydroxypropanoic. At room temperature the molecule is continuously rotating through all possible conformations.
Now let's look at the two most characteristic conformations of ethane, the alternate and eclipsed conformations.
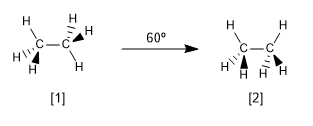
Alternate conformation of ethane
Eclipsed conformation of ethane
Some reactions are highly dependent on the conformation in which the molecule is arranged. Thus, E2 elimination requires that the molecules adopt the ANTI arrangement and its speed is dependent on the concentration of molecules that are in this conformation.
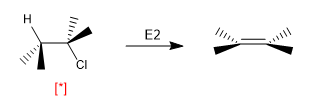
[*] "ANTI" H-Cl arrangement
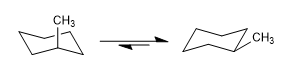
Cyclohexanes also have two chair conformations that interconvert at room temperature. The conformation that leaves more groups in the equatorial position is usually the one with the greatest stability.
Another example of conformational equilibrium is presented by biphenyls. When both rings are arranged in the same plane they form an aromatic system, turning 90º breaks the conjugation between them and makes the molecule more unstable.
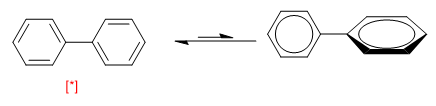
[*] More stable conformation
However, the presence of bulky groups in the ortho positions with respect to both rings produces repulsions that force the molecule to rotate, in this case the conformation with the perpendicular rings is more stable.
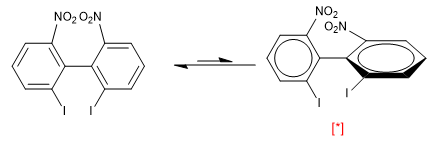
[*] More stable conformation
At room temperature only the most stable conformation is observed (right side). The repulsion between the two nitros and the two iodines is so important that the biphenyl rotation has an activation energy of 19 Kca/mol.
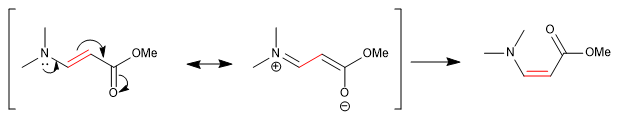
Some links, although apparently simple, do not allow groups to rotate freely around them. DMF methyls give two singlets, at different chemical shifts, in the NMR spectrum because the CN bond has strong double bond character and restricts group rotation.
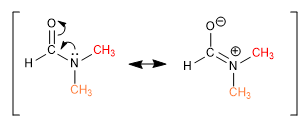
Other bonds, which are apparently double, allow rotation due to the contribution of a resonant structure in which they behave like single bonds.