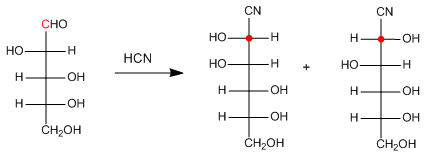
The Kiliani-Fischer synthesis makes it possible to lengthen the monosaccharide chain through the formation and subsequent reduction of cyanohydrins. The big problem of the synthesis is the lack of stereoselectivity.
Stage 1. Formation of cyanohydrin.
Stage 2. Reduction of the nitrile to aldehyde.
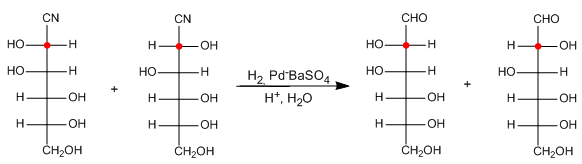
The reduction of the nitrile to aldehyde involves a hydrogenation using a lindlar-type catalyst, which forms an imine. Acid hydrolysis of the imine produces the aldehyde.
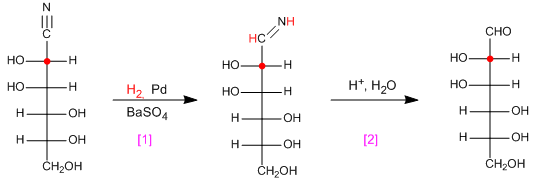
Lindlar catalyst hydrogenation
imine hydrolysis