The rules to give absolute configuration to a stereogenic center are the following:
1. Give priority to the groups that start from the stereogenic center by atomic number
2. In the case of isotopes, priority is given by atomic mass.
3. If the rotation, following the three highest priority groups with the fourth at the bottom, is clockwise, the stereogenic center is R.
4. If the rotation is counterclockwise, the stereogenic center is S.
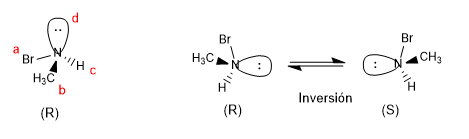
Ammonium salts with four different substituents are chiral compounds, the notation for chiral nitrogen is given the same as for carbon.
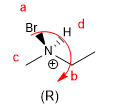
Amines can also be chiral if all three of their substituents are different, the lone pair being the priority group d. However, chiral amines racemize due to nitrogen inversion and the pure enantiomers cannot be isolated.
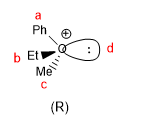
In oxonium ions, oxygen behaves as a sterogenic center, giving rise to chiral molecules.