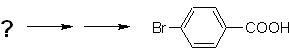SYNTHESIS OF AROMATIC COMPOUNDS I
(Synthesis Tree Method)
Propose a synthesis plan, using toluene or xylene as starting materials, for the following molecules:
(MOb 12)
1-isopropyl-7-methylnaphthalene | (MOb 13)
2,7-dimethylnaphthalene |
(MOb 14)
1-isopropyl-6-methylnaphthalene | (MOb 15)
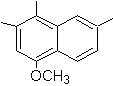
4-methox-1,2,7-trimethylnaphthalene |
Solution: (MOb 12).
In the strategy that is assumed, it is taken into account that the last stage can respond to an "aromatization" process, for which it is proposed that the precursor molecule presents a non-aromatic ring, with a double bond.
on the carbon containing the double bond and the alkyl group.
This structure can be achieved by the action of a Grignard on a carbonyl and the subsequent dehydration of the alcohol formed. The ketone is formed by acylation on the appropriate benzene compound with succinic anhydride and its subsequent Friedel-Crafts intramolecular acylation closures.
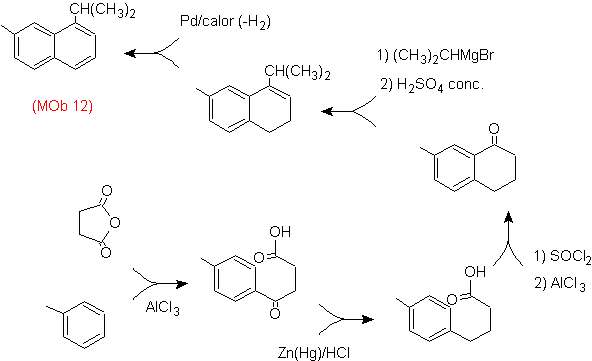
Solution (MOb 13).
Once again, the precursor molecule has to be "aromatized", the most appropriate strategy among others It is based on the combination of acylation with substituted succinic anhydride and the Clemmensen reduction.
The final carbonyl is reduced to alcohol that will then be dehydrated with molecular hydrogen and a catalyst called copper chromyl.
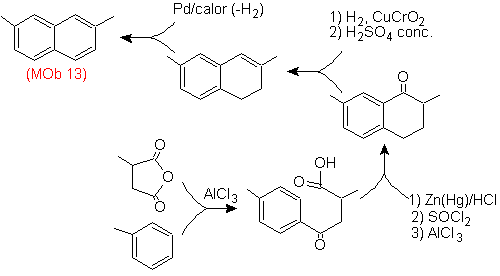
Solution (MOb 14).
The final aromatization is achieved after reducing a carbonyl group to alcohol and then dehydrating it. The precursor molecule is obtained with an acylation with succinic anhydride.
The carbonyl that has to be attacked by a Grignard to introduce the alkyl group and generate the alcohol that will be dehydrated, is temporarily protected as a cyclic acetal.
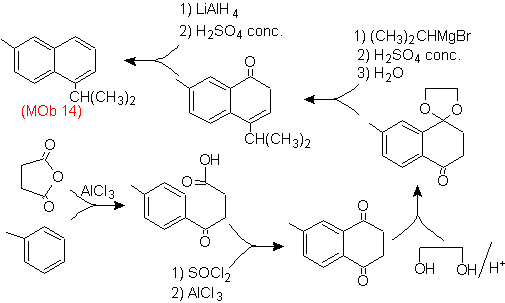
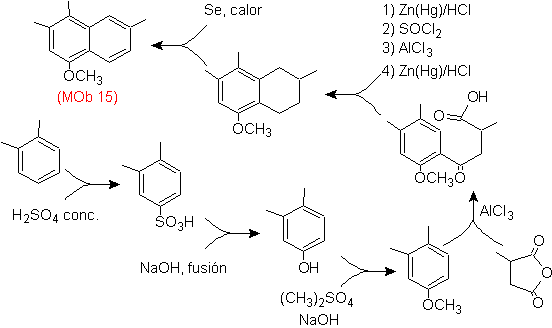
Mob 15 solution .
The most substituted ring is considered to correspond to the aromatic ring that by acylation suitable with a derivative of succinic anhydride, it will allow the closure of a ring, with an intelligent combination of acylation and reduction of the carbonyl.
The benzene ring with the required functionality is prepared from o-xylene, which is sulfonated and the –OH group is protected by etherifying it to the end.
The acylation of naphthalene is subject to an interesting solvent effect. The reaction in carbon disulfide or halogenated solvents produces an attack predominantly in the a position , however, in nitrobenzene solution, the size of the electrophilic attacking agent increases by solvation with nitrobenzene, attacking the b position , which is less impeded sterically.
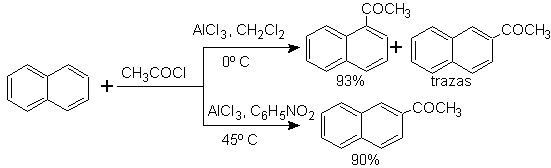
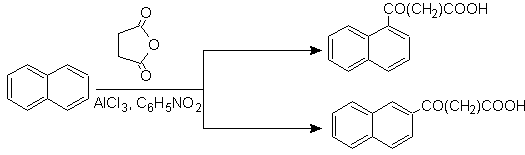
Succinoylation of naphthalene to nitrobenzene produces a separable mixture of a and b isomers , both of which are used in the synthesis of phenanthrene derivatives.
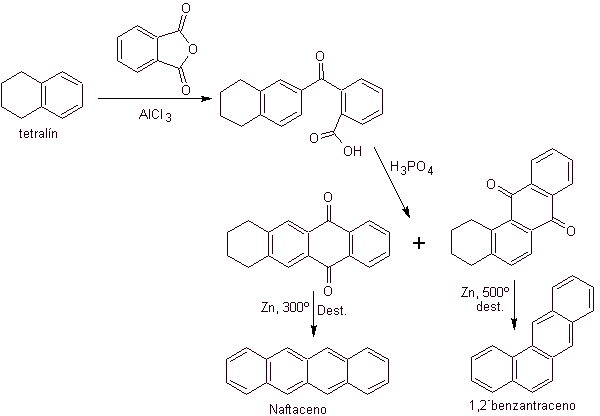
Tetralin is exclusively acylated at the b- position , demonstrating the resistance of 2-phthaloyl derivatives to cyclization at the 3-position to give linear condensates.
Example: Synthesize p -bromobenzoic acid from benzene and the necessary reagents. |
MOb 16 |
Solution (MOb 16).
It is necessary to ask "What is an immediate precursor of p-bromobenzoic acid?" The retrosynthetic analysis of
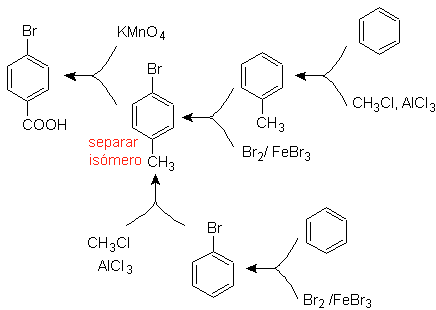
The best option is the one that uses toluene as an intermediate, since the methyl group is weakly activating the benzene ring and improves the yield of p-bromotoluene formation.
In the second synthesis route, it should be taken into account that bromine is ring deactivating, and therefore the yield of the reaction will obviously be much lower than the first route. However, it must be understood that there is not a single product in the synthesis, since the ortho-substituted isomer will also be formed, which must necessarily be separated in the synthesis process.
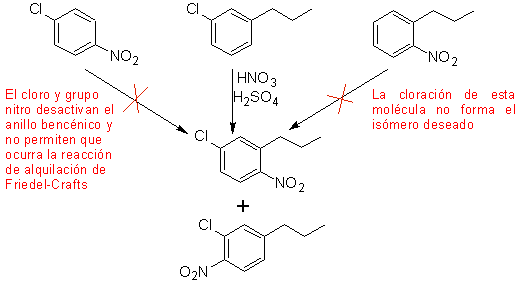
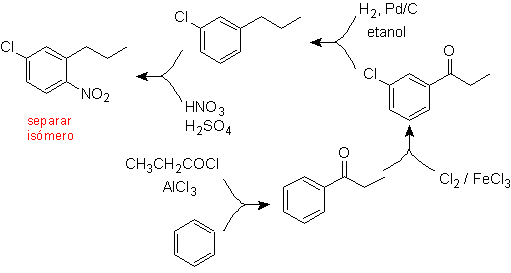
A second case, which shows the characteristics of an electrophilic multi-substitution of the benzene ring, is the synthesis of 4-chloro-1-nitro-2-propylbenzene (MOb. 17) from benzene.
Solution (MOb 17).
In principle there are three possible di-substituted precursors, but only one of them is suitable. Also in this case, there is a high probability of the formation of the 2-chloro-1-nitro-4-propylbenzene isomer.
the ultimate synthesis of
It is now clear that one of the tasks of the chemist is to design synthesis routes that lead as far as possible to the formation of a single product with the highest yields, that is, avoiding the formation of isomers throughout the entire process. To exemplify this aspect, we will study the synthesis of the following molecules:
MOb. 18
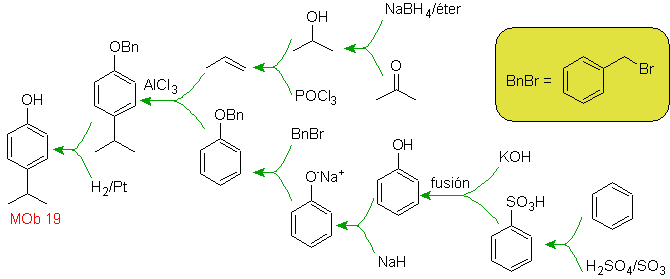
| MOb. 19
|
Solution MOb 18
Bromine is oriented ortho to and also deactivating the benzene ring, so the strategy resorts to placing a strongly activating group at the ortho and para positions of a new electrophile and after generating the reaction, removing the activating group from the ring benzene.
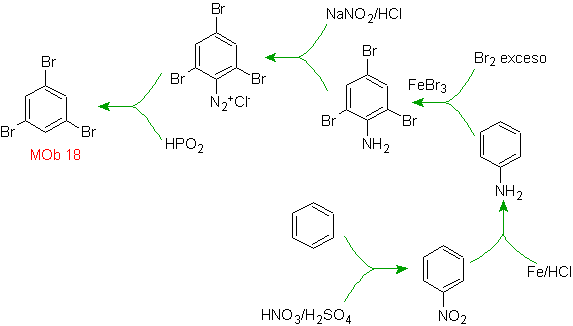
The –OH group is a strong activator of the benzene ring and an ortho-para orientation. Consequently, we can transform the –OH group into a much larger group, that is, bulky, which will exert a steric hindrance for any electrophile that approaches the ring and will orient it only to the para position. Finally, the –OH group will be restored, to reach the target molecule.