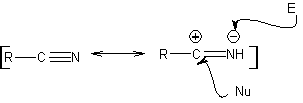
The cyano group in the synthesis of heterocycles
The polarity of the CN bond of the cyano group (also called nitrile) means that this reagent can react with nucleophiles and electrophiles, giving rise to heterocycles with one or several heteroatoms in the ring.

MOb: 75 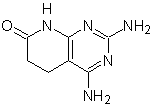 | MOb: 76 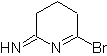 |
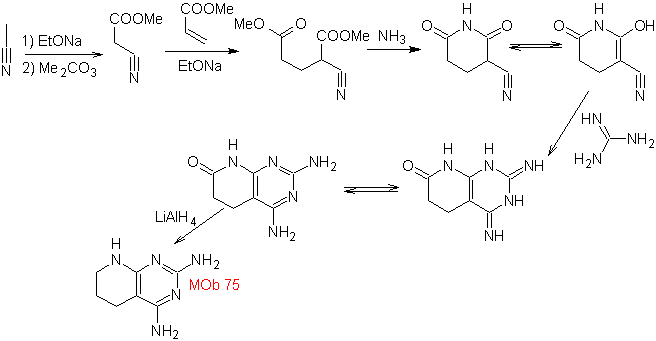
MOb 75 . Retrosynthetic analysis . the MOb , presents in one of the heterocycles, the guanidine structure and in the other heterocycle there is a saturated structure, which can be transformed to a lactam.
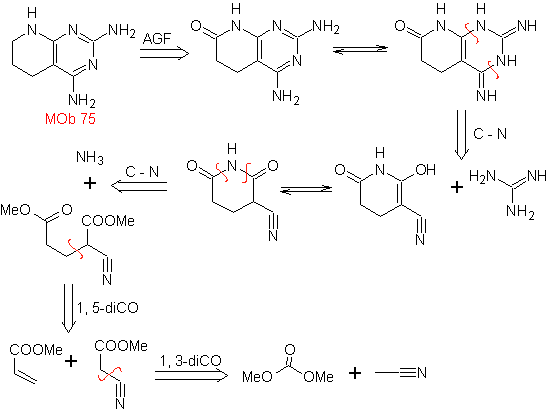
Synthesis : the condensation The aldol type of the starting materials leads to a 1,5-diCO compound, which with NH3 forms the first dilactam cycle capable of combining with guanidine to form the second heterocycle. The CN group is very important in the second cycle.
Finally, the reduction of the C=O groups leads to the MOb 75.
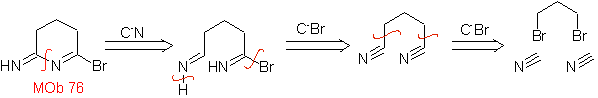
MOb 76. Retrosynthetic analysis . The CN bond of amines is taken into account, for the first disconnection, of the mob 76, then continue with the simultaneous disconnection of an H and a Br.
The planned synthesis operations lead to a precursor molecule with two CN groups, which are disconnected into a dihalide, and two moles of HCN.
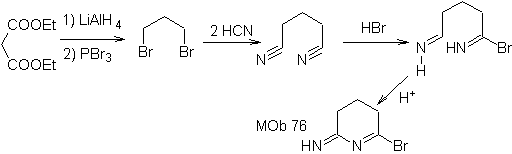
To this system, HBr is added and then, always in an acid medium, cyclization occurs, to form the mob 75